When we think of dementia, we often picture the visible struggles: a person forgetting a familiar face, losing their way on a well-known street, or struggling to find words during a conversation. However, modern neuroscience reveals a much more unsettling reality: the disease is often well underway while the individual remains perfectly sharp, independent, and unaware.
The battle for brain health is not won at the moment of diagnosis; it is fought in the decades of silence that precede it.
The Invisible Phase: Preclinical Alzheimer’s
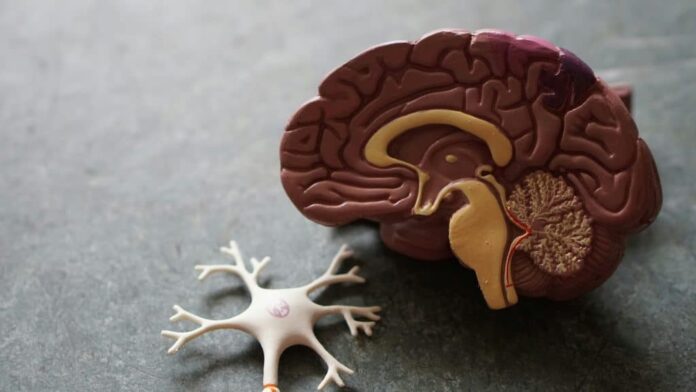
Alzheimer’s disease—which accounts for approximately 70% of all dementia cases—does not begin with memory loss. It begins with biological changes that occur long before the mind shows any signs of decline.
Scientists refer to this as preclinical Alzheimer’s disease. During this stage, the brain is undergoing significant pathological changes, yet the person remains cognitively unimpaired. Current research suggests a staggering scale to this “silent” population:
– The 20-Year Lead Time: Hallmark proteins, such as beta-amyloid, can begin accumulating in the brain as early as two decades before symptoms emerge.
– A Global Scale: It is estimated that as many as 315 million people worldwide may currently be in this preclinical stage.
– The Two-Phase Model: Research indicates a slow, silent first phase that affects specific cell types without causing cognitive symptoms. It is only when the disease enters a second, more aggressive phase—marked by the rapid accumulation of amyloid and tau proteins—that visible damage and memory loss occur.
The tragedy of dementia is the “cruel paradox”: the period when intervention would be most effective is exactly when most people have no idea they are at risk.
Subtle Red Flags: Beyond Memory Loss
While the preclinical stage is largely invisible to standard clinical tests, there is a “bridge” phase known as Mild Cognitive Impairment (MCI). During this time, subtle indicators may appear that are frequently dismissed as “just getting older” or “stress.”
Recognizing these patterns is vital for early detection:
- Emotional & Behavioral Shifts: Apathy (a loss of interest or emotional flattening) and depression are significant indicators. Notably, depression experienced at any point in adulthood has been linked to a doubling of dementia risk later in life.
- Sensory Changes: A declining sense of smell (olfactory loss) and untreated hearing loss are both strongly associated with neurodegenerative decline.
- Sleep & Physical Indicators: Frequent nightmares and disturbed sleep patterns have been linked to higher dementia risk. Additionally, unexplained weight loss can sometimes precede cognitive symptoms due to metabolic changes.
- Language & Cognitive Nuances: Subtle difficulties in finding words or minor lapses in short-term memory (remembering what happened this morning versus decades ago) serve as early warning signs.
The Hope in Prevention: 45% of Cases May Be Avoidable
Perhaps the most significant takeaway from recent neurological research is that dementia is not an inevitable consequence of aging.
The 2024 Lancet Commission on Dementia Prevention reported that approximately 45% of all dementia cases worldwide could potentially be prevented by addressing 14 modifiable risk factors. This highlights a shift in how we view the disease: it is increasingly seen as a condition with deep roots in lifestyle, environment, and social factors.
Key Areas for Intervention:
- Midlife Health (Ages 18–65): This is the most critical window for cumulative impact. Managing hypertension, obesity, diabetes, and hearing loss, while avoiding smoking and excessive alcohol, can significantly protect the brain.
- Later Life (Ages 65+): Addressing social isolation, managing vision loss, and controlling high LDL cholesterol are vital protective measures.
- Lifelong Habits: Regular physical activity, cognitive stimulation, and maintaining high educational attainment provide a foundation of “cognitive reserve.”
The Window of Opportunity
The goal of modern medicine is to “move upstream.” Rather than waiting for irreversible damage to occur, researchers are focusing on the preclinical window.
New technologies—such as blood-based tests for amyloid and tau proteins, advanced MRI modeling, and the recent FDA approval of anti-amyloid immunotherapies (like lecanemab)—are designed to catch the disease during its silent phase. If we can intervene while the brain is still functionally connected, we may be able to slow or even delay the onset of symptoms entirely.
Conclusion
Dementia is a slow-moving thief that begins its work decades before it is detected. However, by focusing on midlife health and recognizing subtle early warning signs, we can move from reactive treatment to proactive prevention, potentially reclaiming much of the territory the disease seeks to take.